FDA Science: Childhood Vaccine Clinical Trial Series
Compilation of 17 part series of tweets from Aaron Siri
Last month Aaron Siri did an incredible series of 17 tweets on childhood vaccine clinical trials. All summarized and referenced. Aaron also has two excellent related articles in his Injecting Freedom substack which further discuss the lack of placebo controls:
Clinical Trial to License RotaTeq, Like Almost All Childhood Vaccines, Did Not Use a Placebo Control
What the “Casual Cruelty” of Dr. Paul Offit Reveals — the last section of this article titled “Proof Regarding the Clinical Trials Relied Upon by the FDA to License the Childhood Vaccines on the CDC Childhood Vaccine Schedule” also contains a list of the childhood vaccines and clinical trial information. There is considerable overlap with the tweet series but there is also some unique info in both.
I wanted this information handy for reference in addition to the articles and thought you might find it helpful too. I so appreciate the work that Aaron Siri and his team put into both the articles and the series of tweets!
FDA Science: Childhood Vaccine Clinical Trial Series (Part 1 of 17)
https://twitter.com/AaronSiriSG/status/1683914950975291394
“The clinical trials for Hepatitis B vaccines, injected at birth and again at 1 and 6 months, showed they were safe to license!” It’s misinformation to claim otherwise!
—Recombivax HB (Merck) licensed for babies based on trials with no placebo control & 5 days of safety monitoring after injection.
--Engerix B (GSK) licensed for babies based on trials with no placebo control & 4 days of safety monitoring after injection.
Sources: https://fda.gov/media/74274/download… (Recombivax HB package insert, see Section 6.1); https://fda.gov/media/119403/download… (Engerix B package insert, see Section 6.1); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 2 of 17)
https://twitter.com/AaronSiriSG/status/1683962124601806848
“The clinical trials for DTaP vaccines, injected into babies at 2, 4, 6 and 15 months and again at 4 years of age, showed they were safe to license!” It’s misinformation to claim otherwise!
--Infanrix (GSK) licensed for babies based on trials with no placebo control (DTP vaccine used as a control) & up to 30 days of safety review after injection.
--Daptacel (Sanofi) licensed for babies based on trials with no placebo control (DT or DTP vaccine used as control) & 2 months of safety review after injection except one trial which was 6 months with no control, 1,454 children and “[w]ithin 30 days following any dose of DAPTACEL, 3.9% subjects reported at least one serious adverse event.” Note that DTP vaccine (used as the control in DTaP vaccine trials) was not tested in a placebo-controlled trial and studies roundly show DTP vaccine increases overall mortality, see https://icandecide.org/wp-content/uploads/2021/06/2021.01.28-Letter-to-Special-Rapporteur-on-Poverty.pdf…
Sources: https://fda.gov/media/75157/download… (See Section 6.1 of Infanrix package insert); https://fda.gov/media/74035/download… (See Section 6.1 of Daptacel package insert); https://fda.gov/safety/reporting-serious-problems-fda/what-serious-adverse-event… (FDA definition of “serious adverse event”); https://cdc.gov/vaccines/schedules/…hcp/imz/child-adolescent.html (childhood vaccine schedule).
FDA Science: Childhood Vaccine Clinical Trial Series (Part 3 of 17)
https://twitter.com/AaronSiriSG/status/1683991697750114305
“The clinical trials for PCV (Pneumococcal conjugate) vaccines, injected into babies at 2, 4, 6 and 12 months, showed they were safe to license!” It’s misinformation to claim otherwise!
--Prevnar 13, PCV-13 (Wyeth, part of Pfizer) licensed for babies based on trials with no placebo control (Prevnar 7 used as a control, and Prevnar 7 was licensed based on trial in which the control was another experimental vaccine) & 6 months of safety review after injection which found, “Serious adverse events reported following vaccination in infants and toddlers occurred in 8.2% among Prevnar 13 recipients and 7.2% among Prevnar 7 recipients.”
--Vaxneuvance PCV-15 (Merck) licensed for babies based on trials with no placebo control (Prevnar 13 used as the control) & up to 6 months of safety review after injection finding that, “Among children who received VAXNEUVANCE (N=3,349) or Prevnar 13 (N=1,814) … serious adverse events up to 6 months following vaccination with the 4-dose series were reported by 9.6% of VAXNEUVANCE recipients and by 8.9% of Prevnar 13 recipients.” Deemed “safe” because, “[t]here were no notable patterns or numerical imbalances between vaccination groups.”
--Prevnar 20, PCV-20 (Pfizer) licensed for babies based on trials with no placebo control (Prevnar 13 was used as the control) & up to 6 months of safety review after injection that again showed high rates of serious events (this time broken up into two categories – “serious adverse events (SAEs)” and “newly diagnosed chronic medical conditions (NDCMCs)”) in both vaccine groups but deemed “safe” because “no notable patterns or imbalances between vaccine groups.”
Sources: https://fda.gov/media/76076/download… (See Adverse Reactions section of Prevnar 7 package insert stating the control in the clinical trials for Prevnar 7 was an “Investigational meningococcal group C conjugate vaccine”); https://fda.gov/media/107657/download… (See Section 6.1 of Prevnar 13 package insert); https://fda.gov/media/150819/download… (See Section 6.1 of Vaxneuvance package insert); https://fda.gov/media/149987/download… (See Section 6.1 of Prevnar 20 package insert); https://fda.gov/media/150459/download?attachment… (See Clinical Review Memo, June 8, 2021, for Prevnar 20 discussing SAEs and NDCMCs); https://fda.gov/safety/reporting-serious-problems-fda/what-serious-adverse-event… (FDA definition of “serious adverse event”); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 4 of 17)
https://twitter.com/AaronSiriSG/status/1684011163678576641
“The clinical trials for Inactivated poliovirus vaccine, injected into babies at 2, 4, and 6 months and again at 4 years of age, showed it was safe to license!” It’s misinformation to claim otherwise!
--IPOL (Sanofi) licensed in 1990 for babies based on trials with no placebo control & 3 days of safety review after injection. Sanofi reports that, “Although no causal relationship has been established, deaths have occurred in temporal association after vaccination of infants with IPV.”
IPOL is an injected polio vaccine and is the only polio vaccine used in the U.S. for over two decades. It is a different product than the polio vaccine developed by Jonas Salk in the 1950s (and ceased being used in the U.S. in the 1960s). Unlike Salk’s vaccine, “IPOL is grown in vero cells, a continuous line of monkey kidney cells cultivated on microcarriers,” with a different “culture technique,” different “purification, concentration, and standardization,” and contains 40, 8 and 32 versus Salk’s 20, 2 and 4 D antigen units of PV types 1, 2, and 3. Hence, the trials of Salk’s vaccine from the early 1950s were not relied upon to license IPOL.
Sources: https://fda.gov/media/75695/download… (See “Description” section and “Adverse Reactions” section of IPOL package insert); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 5 of 17)
https://twitter.com/AaronSiriSG/status/1684307622722412545
“The clinical trials for Hib (Haemophilus influenza type b) vaccines, injected into babies at 2, 4, 6 and 12 months, showed they were safe to license!” It’s misinformation to claim otherwise!
--ActHIB (Sanofi) licensed for babies based on trials with no placebo control (Hepatitis B vaccine used as control) & 30 days of safety review after injection during which 3.4% experienced a serious adverse event but “[n]one was assessed by the investigators [Sonafi] as related to the study of vaccines.”
--Hiberix (GSK) licensed for babies based on trials with no placebo control (Unlicensed Hib vaccines and HibTITER used as the control) & 31 days of safety review after injection.
--Liquid PedvaxHIB (Merck) licensed for babies based on trials with no placebo control (Lyophilized PedvaxHIB used a control) & 3 days of safety review after injection.
Sources: https://fda.gov/media/74395/download… (See Section 6.1 of ActHIB package insert); http://wayback.archive-it.org/7993/20170723144656/https:/www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM244597.pdf… (See Safety section of FDA’s Summary Basis of Approval for ActHIB); https://fda.gov/media/77017/download… (See Section 6.1 of Hiberix package insert); http://wayback.archive-it.org/7993/20170722072902/https://www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM182550.pdf… (FDA Clinical Review for Hiberix describing controls used); https://fda.gov/media/80438/download… (See Section 6.1 of Liquid PedvaxHIB package insert describing that the control was Lyophilized PedvaxHIB and that vaccine was tested in a trial in which controls were given placebo, OPV and DTP but there is no indication Lyophilized PedvaxHIB was ever licensed); https://fda.gov/safety/reporting-serious-problems-fda/what-serious-adverse-event… (FDA definition of “serious adverse event”); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule
FDA Science: Childhood Vaccine Clinical Trial Series (Part 6 of 17)
https://twitter.com/AaronSiriSG/status/1684325221745184768
“The clinical trials for Rotavirus vaccines, given by oral drops to babies at 2, 4 and 6 months, showed they were safe to license!” It’s misinformation to claim otherwise!
--Rotarix (GSK) licensed for babies based on trials without a placebo control (the control group received an oral drop that included Dextran, Sorbitol, Amino Acids, Dulbecco’s Modified Eagle Medium, and Xanthan) & 31 days of safety review after oral dose and up to a year in some trials for cases of intussusception. There were more deaths in the group receiving Rotarix than the purported placebo. As disclosed by the FDA and GSK: “During the entire course of 8 clinical studies (Studies 1 to 8), there were 68 (0.19%) deaths following administration of ROTARIX (n = 36,755) and 50 (0.15%) deaths following placebo administration (n = 34,454). The most commonly reported cause of death following vaccination was pneumonia, which was observed in 19 (0.05%) recipients of ROTARIX and 10 (0.03%) placebo recipients (RR: 1.74, 95% CI: 0.76, 4.23).”
--RotaTeq (Merck) licensed for babies based on trials without a placebo control (the control group received an oral drop that included Polysorbate-80 and Sodium Phosphate) & 42 days of safety review after each oral dose and up to a year for cases of intussusception. Note that every vaccine on the CDC childhood schedule is given via injection, except for one flu vaccine given by nasal spray and the rotavirus vaccines, which are given by oral drops in the mouth.
Sources:
https://fda.gov/media/163009/download… (Package insert for Rotarix claiming “placebo” was used in the clinical trial);
http://wayback.archive-it.org/7993/20170722073219/https:/www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM133580.pdf…(Clinical review of Rotarix admitting that the “The placebo consisted of all components of Rotarix, but without any RV particles” which means it included, among other ingredients, Dextran, Sorbitol, Amino Acids, Dulbecco’s Modified Eagle Medium, and Xanthan); https://fda.gov/media/75718/download…(Package insert for RotaTeq claiming “placebo” was used in the clinical trial);
https://icandecide.org/wp-content/uploads/2023/06/rotateq_placebo.pdf… (See page 1788 of the Clinical Study Report for RotaTeq explaining the purported “placebo” actually included Polysorbate-80 and Sodium Phosphate); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 7 of 17)
https://twitter.com/AaronSiriSG/status/1684329799781863424
“The clinical trials for Pfizer’s Covid-19 vaccine, injected into babies at 6, 7 and 10 months, showed it was safe to license!” It’s misinformation to claim otherwise!
--Comirnaty (Pfizer) licensed for babies based on trial with a placebo control (finally!) & 6 months of safety review after injection.
Note that Comirnaty (Pfizer) is currently only licensed for 12 years and older and Spikevax (Moderna) is only licensed for 18 years and older. Also, while Comirnaty’s trial had a placebo control group, that group was unblinded and most were vaccinated during the 6-month safety review period. The 16- and 17-year-old data is not separated from the adult data, but the 12- to 15-year-old data is separated and included only 1,131 children who received a vaccine, and the case of Maddie de Garay reflects how this trial was conducted.
Sources: https://fda.gov/media/151707/download… (See Section 6.1 of the Comirnaty package insert); https://thehighwire.com/ark-videos/rigged-maddies-story/…(video regarding Maddie de Garay); https://icandecide.org/3-08-2022-ltr-to-dr-paul-richards-fda-re-maddie-de-garay/…(documents related to Maddie de Garay); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 8 of 17)
https://twitter.com/AaronSiriSG/status/1684340015269306370
“The clinical trials for Influenza vaccines, injected into babies at 6 and 7 months, and then again annually, showed they were safe to license!” It’s misinformation to claim otherwise!
The formulation for each influenza vaccine changes annually and there is no clinical trial carried out for each new formulation.
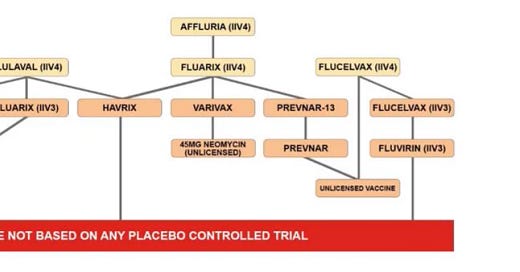
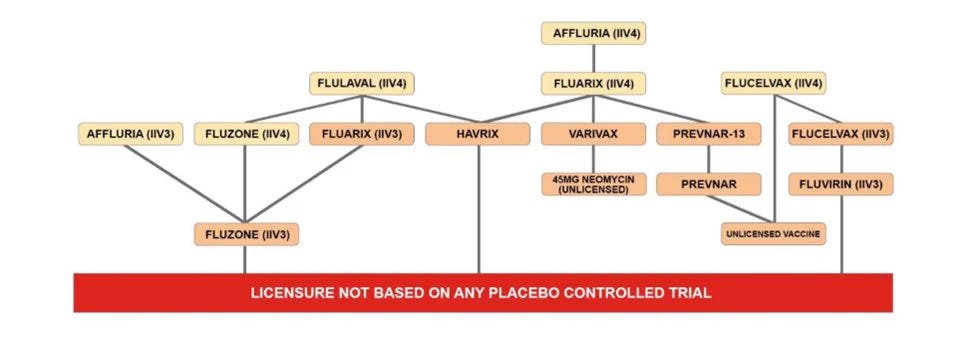
In any event, none of the clinical trials for the original formulation of any injected influenza vaccine for children had a placebo control group (see chart below). The fact that a placebo control could and should have been used is clear from the fact that a placebo control was used for some of the influenza vaccine trials for adults. Also, there is one inhaled influenza vaccine that had a placebo control but, again, its formulation changes every year and is not safety tested in any trial.
Sources: https://cdc.gov/flu/professionals/acip/2022-2023/acip-table.htm… (Table of influenza vaccines licensed for 2022-2023 influenza season and age indication for each); https://fda.gov/vaccines-blood-biologics/vaccines/vaccines-licensed-use-united-states… (See the “Children” portion of Section 6.1 for each influenza vaccine indicate for use in children); https://icandecide.org/wp-content/uploads/2019/09/ICAN-Reply-1.pdf… (see page 13 and 14 detailing each influenza vaccine with FDA citations to support each data point in the chart below)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 9 of 17)
https://twitter.com/AaronSiriSG/status/1684344319904448517
“The clinical trials for MMR (measles, mumps, rubella) vaccines, injected at 12 months and 4 years of age, showed it was safe to license!” It’s misinformation to claim otherwise! -
-M-M-R-II (Merck) licensed based on a trial with no placebo control & 42 days of safety review after injection in a trial with a total of only 834 children of which a third developed gastrointestinal issues and a third respiratory issues.
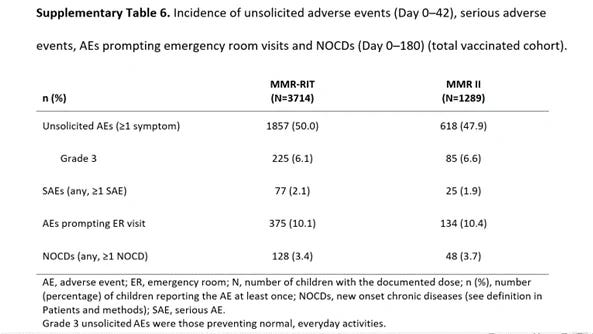
--Priorix (GSK) licensed based on trials with no placebo control (M-M-R-II used as the control) & 6 months of safety review after injection in which both vaccine groups had a high rate of serious adverse events, emergency room visits, and new onset of chronic diseases (e.g., autoimmune disorders, asthma, type I diabetes, vasculitis, celiac disease, thrombocytopenia, and allergies) as reflected in the chart below.
Source: https://sirillp.com/MMRII-FOIA (Clinical trials reports to license M-M-R-II); https://fda.gov/media/158941/download…(See Section 6.1 of Priorix package insert which fails to discuss serious adverse events, emergency room visits, and new onset chronic health conditions occurring in the trial); https://ncbi.nlm.nih.gov/pmc/articles/PMC7192400/bin/piz010_suppl_supplementary_materials.docx…(See Table 6 of the Supplementary Materials for Priorix); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 10 of 17)
https://twitter.com/AaronSiriSG/status/1684356948010110976
“The clinical trials for Varicella (Chicken Pox) vaccine, injected at 12 months and 4 years of age, showed it was safe to license!” It’s misinformation to claim otherwise!
--Varivax (Merck) licensed based on trials with no placebo control (the purported “placebo” was actually an injection of 45 mg of neomycin per milliliter) & 70 days of safety review after injection which included only one controlled trial of 956 children in which around half received Varivax and half received the injection of 45 mg of neomycin per milliliter, and there was one trial in which 32 children received Varivax and 29 children received nothing and then received Varivax eight weeks later; during this eight-week period, the Varivax group had double the rate of ear infection and a 50% increase in respiratory infection. As for serious adverse events, Merck did not consider any related to Varivax.
Source: https://fda.gov/media/76000/download…(See Section 6.1 of the package insert for Varivax); https://pubmed.ncbi.nlm.nih.gov/6325909/(Merck study explaining the “placebo” was in fact an injection of neomycin); https://sirillp.com/Varivax-clinical-trials… (clinical trial reports for Varivax); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 11 of 17)
https://twitter.com/AaronSiriSG/status/1684364751676272641
“The clinical trials for Hepatitis A vaccines, injected at 12 months and 18 months of age, showed they were safe to license!” It’s misinformation to claim otherwise!
--Havrix (GSK) licensed based on trials with no placebo control (Engerix-B was used as a control) & 31 days of safety review after injection with a phone call follow-up at 6 months.
--Vaqta (Merck) licensed based on trials with no placebo control (an injection of AAHS, an aluminum adjuvant, and thimerosal, a form of mercury, were used as a control) & up to 42 days of safety review after injection.
Clinical trials for both these products occurred at roughly the same time and, because there was no licensed Hepatitis A vaccine at that time, there was no excuse for not using a placebo control in the above trials. It is also startling that Engerix-B, which had 4 days of safety monitoring in its trial, was used as the control for Havrix, and that an injection of known cyto-and-neuro toxic substances, AAHS and thimerosal, were used as a control for Vaqta instead of just a saline injection.
Source: https://fda.gov/media/119388/download…(See Section 6.1 of the package insert for Havrix); https://fda.gov/media/74519/download…(See Section 6.1 of the package insert for Vaqta describing the control as “placebo (alum diluent)”); https://nejm.org/doi/full/10.1056/NEJM199208133270702…(See Merck study reflecting the control in the Vaqta trial was not a placebo because it contained 300µg AAHS and thimerosal); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 12 of 17)
https://twitter.com/AaronSiriSG/status/1684371561011384320
“The clinical trials for Tdap (tetanus, diphtheria, & acellular pertussis) vaccines, injected at 11 years of age, showed they were safe to license!” It’s misinformation to claim otherwise!
--Adacel (Sanofi) licensed based on trials with no placebo control (Td, for adult use, was used as a control) & up to 6 months of safety review after injection. -
-Boostrix (GSK) licensed based on trials with no placebo control (DECAVAC or Adacel was used as a control) & up to 6 months of safety review after injection.
Source: https://fda.gov/media/119862/download…(See Section 6.1 of the package insert for Adacel); https://fda.gov/media/124002/download…(See Section 6.1 of the package insert for Boostrix); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule)
FDA Science: Childhood Vaccine Clinical Trial Series (Part 13 of 17)
https://twitter.com/AaronSiriSG/status/1684373445885456384
“The clinical trials for HPV (human papillomavirus) vaccine, injected at 9 and 9½ years of age, showed it was safe to license!” It’s misinformation to claim otherwise!
--Gardasil 9 (Merck) was licensed based on trials in which safety was reviewed after injection for 1 month in five of the clinical trials, 6 months in a lot consistency trial, and 4 years in one trial of women aged 16 to 26 years (reflecting that a safety trial of a more appropriate duration is possible). These Gardasil 9 trials were either not controlled or used Gardasil 4 as the control except for one trial in which 306 participants received a placebo (but only after receiving the full series of Gardasil 4 injections).
Merck states that in the Gardasil 9 clinical trial, “2.2% of GARDASIL 9 recipients and 3.3% of GARDASIL 4 recipients reported new medical conditions potentially indicative of systemic autoimmune disorders.” In the Gardasil 4 clinical trial, the control was an injection of aluminum adjuvant and 2.3% of the Gardasil group and 2.3% of the aluminum adjuvant control group had a suspected autoimmune disorder (and aluminum adjuvant is used to induce autoimmunity in lab animals).
Source: https://wayback.archive-it.org/7993/20190423065200/https:/www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM429166.pdf…(See pages 17 to 19 of the FDA clinical review for Gardasil 9 which provides a chart summarizing the trials); https://fda.gov/media/74350/download…(See Table 9 of package insert for Gardasil 4, noting that of the 9,412 combined “AAHS Control or Saline Placebo” participants, at most 320 were in the “Saline Placebo” group which actually included all Gardasil ingredients except for the antigens and the AAHS); https://pubmed.ncbi.nlm.nih.gov/27417999/(Study inducing autoimmunity in lab animals with aluminum adjuvant); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule).
FDA Science: Childhood Vaccine Clinical Trial Series (Part 14 of 17)
https://twitter.com/AaronSiriSG/status/1684381232459821056
“The clinical trials for MenACWY (meningococcal) vaccines, injected at 11 and 16 years of age, showed they were safe to license!” It’s misinformation to claim otherwise!
--Menactra (Sanofi) licensed based on trials with no placebo control (Menomune used as the control, and amazingly the safety section of the package insert for Menomune lists this same trial in which it is being used as a control) & up to 6 months of safety review after injection.
--Menveo (GSK) licensed based on trials with no placebo control (Menactra, Boostrix, or other vaccines used as a control) & up to 6 months of safety review after injection.
--MenQuadfi (Sanofi) licensed based on trials with no placebo control (Menveo or other vaccines used as a control) & up to 6 months of safety review after injection. The foregoing is another good example of the pyramid of safety in which Menomune is licensed without a placebo control trial and is then used as the control to license Menactra, then Menactra is used as the control to license Menveo, and then Menveo is used as the control to license MenQuadfi. What is the actual safety profile, putting aside the limited six-month safety review period, remains unknown since Menomune is used as the baseline for safety, despite never establishing its safety in a placebo-controlled clinical trial.
Source: https://fda.gov/media/83562/download…(See Section 6.1 of the package insert for Menomune); https://fda.gov/media/75619/download…(See Section 6.1 of the package insert for Menactra); https://fda.gov/media/78514/download…(See Section 6.1 of the package insert for Menveo); https://fda.gov/media/137306/download…(See Section 6.1 of the package insert for MenQuadfi); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule).
FDA Science: Childhood Vaccine Clinical Trial Series (Part 15 of 17)
https://twitter.com/AaronSiriSG/status/1684387211071078403
“The clinical trials for MenB (meningococcal serogroup B) vaccines, injected starting at 10 years of age to certain populations, showed they were safe to license!” It’s misinformation to claim otherwise!
--Bexsero (GSK) licensed based on trials with no placebo control group (either uncontrolled or control group was given an injection of aluminum hydroxide and, in one trial involving 120 adolescents, a saline injection followed by an injection of Menveo and hence FDA labels this an “active control” and not a “placebo control” trial) & 30 days of safety review after injection.
--Trumenba (Pfizer) licensed based on trials with no placebo control group other than 12 people in a dose ranging phase II study (otherwise the controls were injection of Gardasil+placebo, dTaP-IPV+placebo, HepA+placebo, or Menactra+Adacel+placebo) & 30 days of safety review after injection for one of the three trials and up to 11 months in the other two trials.
Source: https://wayback.archive-it.org/7993/20190425012223/https://www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM434748.pdf…(See pages 14 and 15 of FDA Summary Basis of Regulatory Action for Bexsero outlining the clinical trials); https://wayback.archive-it.org/7993/20190423064855/https://www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM434714.pdf…(See page 40 of the FDA Clinical Review for Bexsero admitting the purported “Placebo” included aluminum hydroxide); https://wayback.archive-it.org/7993/20190425012035/https://www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM548305.pdf…(See page 4 of FDA Summary Basis of Regulatory action for Trumenba describing the trials completed after accelerated approval); https://wayback.archive-it.org/7993/20190423065758/https://www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM424626.pdf…(See pages 9 and 10 of the FDA Clinical Review for Trumenba describing the initial trials relied upon for licensure); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule).
FDA Science: Childhood Vaccine Clinical Trial Series (Part 16 of 17)
https://twitter.com/AaronSiriSG/status/1684387423399325696
“The clinical trials for PPSV23 (pneumococcal polysaccharide) vaccine, injected starting at 2 years to certain populations, showed it was safe to license!” It’s misinformation to claim otherwise!
--Pneumovax 23 (Merck) is licensed for children 2 years and older but there is no indication that there was any clinical trial involving anyone younger than 16 years of age that the FDA relied upon to license this vaccine. The package insert and FDA documentation regarding licensure of this product is included in the link below.
Source: https://fda.gov/vaccines-blood-biologics/vaccines/pneumovax-23-pneumococcal-vaccine-polyvalent…(FDA documentation regarding licensure of Pneumovax 23); https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule).
FDA Science: Childhood Vaccine Clinical Trial Series (Part 17 of 17)
https://twitter.com/AaronSiriSG/status/1684387982781054976
The Dengue vaccine is the 17th and last vaccine on the CDC’s childhood vaccine schedule, and the first vaccine that apparently underwent a longer-term placebo-controlled trial prior to licensure! This trial stands as the proof that a longer-term placebo-controlled trial of a childhood vaccine is possible!--Dengvaxia (Sanofi) licensed based on a trial with 11,474 children receiving a placebo control (saline injection) & 5 years of safety review after injection.
For this vaccine, it was learned that children under 6 years old had an increased risk of severe harm and death from this vaccine – harm that would likely never be uncovered by the trials performed for any of the other 16 vaccines in this series. It was also found that children older than 6 who had never had dengue and received this vaccine likewise had a seriously increased risk of severe harm and death. Hence, this vaccine is only to be given to older children who have previously had dengue. As disclosed by the FDA and Sanofi: “Those not previously infected are at increased risk for severe dengue disease when vaccinated and subsequently infected with dengue virus.” This vaccine is only recommended for children in endemic dengue areas and dengue is not endemic in the United States.
Source: https://fda.gov/media/125481/download…(See page 10 of the FDA Statistical Review for Dengvaxia summarizing the clinical trials for this vaccine and see page 1 for disclaimer regarding harm it can cause in children not previously infected with dengue); https://fda.gov/media/124379/download…(See Warnings and Precautions stating “DENGVAXIA is not approved for use in individuals younger than 6 years of age. In persons younger than 6 years of age regardless of previous infection by dengue virus, an increased risk of severe and hospitalized dengue disease can occur following vaccination”); https://usgs.gov/faqs/what-constitutes-united-states-what-are-official-definitions…; https://cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html… (CDC childhood vaccine schedule providing Dengvaxia is only for children “Seropositive [meaning they previously had dengue] in endemic dengue areas”).


Thank you SO for your article - powerful, informative & disturbing. There has never been a safe & effective vaccine, EVER. 'Turtles all the Way Down' powerful book ...